Background: Chimeric antigen receptor T-cell (CAR-T) therapy has emerged as a remarkably efficacious treatment modality in recent years for refractory and relapsed hematopoietic malignancies. However, the exorbitant cost and intricate manufacturing procedure of ex vivo CAR-T cell generation have impeded its broader application. In vivo induced CAR-T therapy offers the potential to circumvent cumbersome manufacturing logistics, thereby providing novel insights into the field of CAR-T treatment. In this study, we have developed a bio-orthogonally redirected bispecific lentiviral vector platform for the reprogramming of circulating T lymphocytes into CAR-T cells and subsequent elimination of tumor cells in vivo.
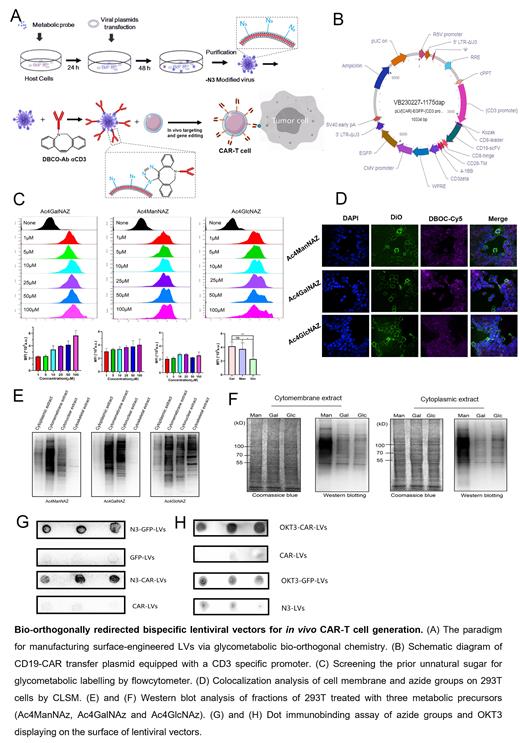
Methods: To engineer dual-targeting lentiviral vectors (D-LVs), the packing cell lines 293T was labelled with azide groups by glycometabolic bio-orthogonal chemistry (designated N 3-293T) firstly, and a CD3 promoter (CD3p) was fused to CAR transfer plasmid. Secondly, azide groups modified LVs were further surface engineered with anti-CD3 antibody (OKT3) via click chemistry. The conjunction of OKT3 and was analyzed using dot immunobinding assay, confocal microscopy and the in vitro transduction efficiency was evaluated using flow cytometry. To demonstrate the ability of targeted transduction and specific cytotoxicity in vivo, D-LVs were intravenously infused into humanized NOD-scid-IL2Rγnull (huNSG) mice engrafted with Nalm6-luc cells. Subsequently, the tumor burden was monitored using a noninvasive bioluminescence imaging system and the in-vivo CD19 -CAR-T cells existence were detected by flow cytometry.
Results: By displaying OKT3 on the single lentiviral surface and fused CD3p into the key construct, we could achieve targeted delivery of CD19-CAR genes to T cells both in vitro and in vivo.
Conclusion: These results demonstrate the great potential applications of this engineered lentiviral system as a new strategy for inducing CAR-T immunotherapy in vivo and a promising approach for leading personalized treatment.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal